Hard Biology: Toxicity
There's a million reasons why biology is hard tech. Today we're talking about one of them: toxicity
Transcript
Let's take a minute to appreciate engineered cells and everything they do for us. You've got your microbes out there making food proteins, flavor molecules, enzymes, industrial chemicals. You've got your mammalian cells out there making biologics, delivery capsids for gene therapy, or even trying to do it all themselves as a cell therapy.
At Ginkgo, we engineer these cells to support our customers's R&D projects. Sometimes you've got to wonder: how do the cells feel about all this? So I asked them, and they said: "Jake, we're cells, we don't have human feelings." But then they said: "Our biggest complaint is toxicity."
Toxicity is the word I'm using to mean anything that cells die faster or grow slower. Sick cells tend to have lower performance in all the ways that you might imagine. They make less of your product. They're less robust to stress or changing conditions. They're just kind of blah and that's not fun.
In a way, it's remarkable that toxicity isn't a bigger problem. After 3.7 billion years of evolution, cells are tough little bastards. They're naturally equipped with any number of homeostasis mechanisms that maintain their core functions stable and buffered against perturbation.
Still, we push them pretty hard. We want the maximum production of proteins, enzymes, or small molecules from our bioprocess. Sometimes, toxicity happens because we're at a limit. In other cases, toxicity is just an inevitable part of engineering a complex system. Things can break in unexpected ways.
I'll break it down into a few common types:
The metabolic burden of gene expression
Damage caused by protein misfolding and aggregation
Toxic metabolic intermediates and finally
Toxic products
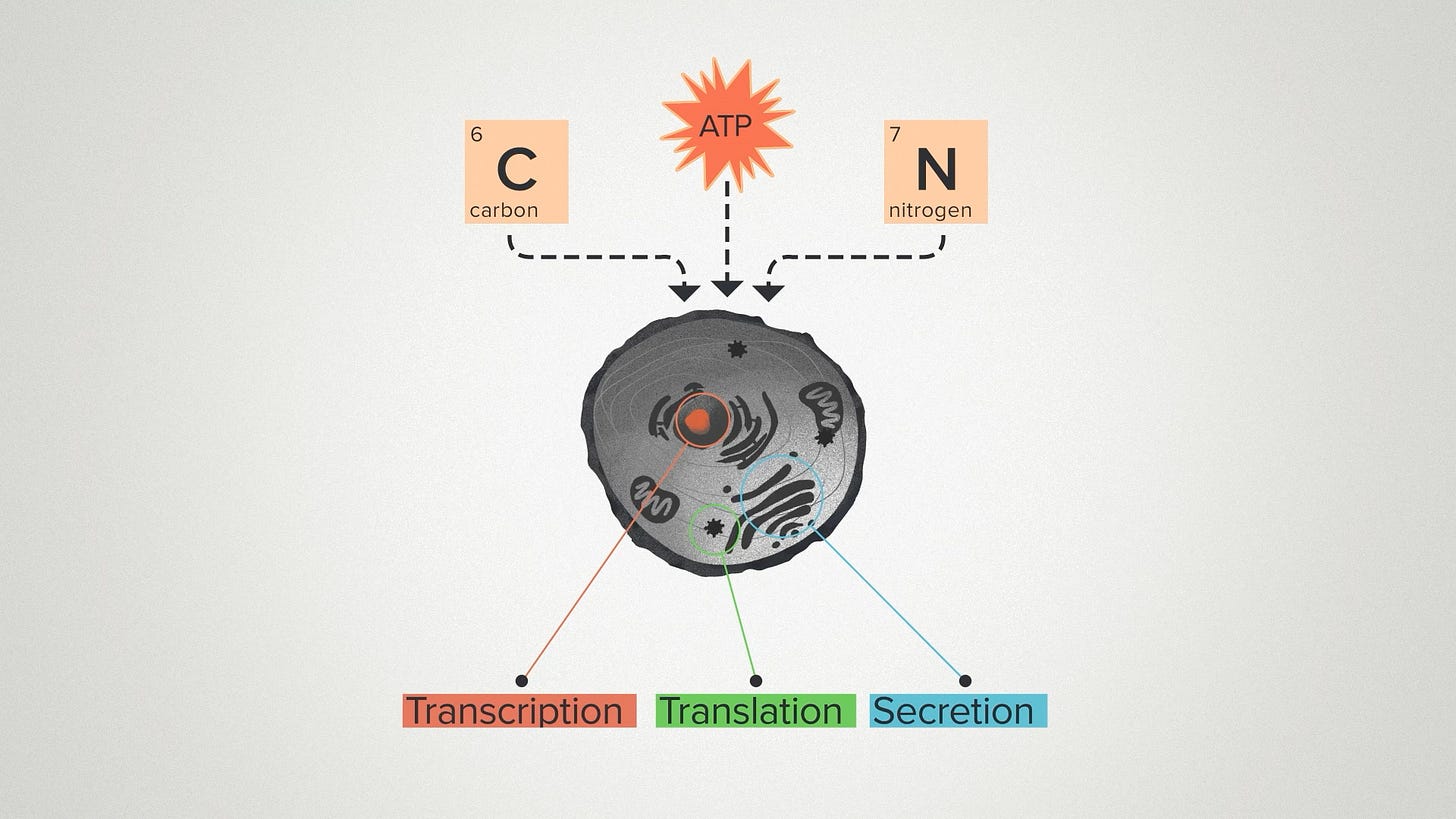
The metabolic burden is the inescapable fact that anything we add to the cell takes resources. The energy and nutrients that go into making your product are not going into growing and maintaining the cell.
In any given project, some of the resources inside a cell are going to be more limiting than others. It could be raw inputs like energy, carbon or nitrogen sources. It could be the use of the cellular machinery for transcription, translation, or secretion. So minimizing the metabolic burden is a balancing act, fine tuning our designs to use cellular resources in the most efficient way.
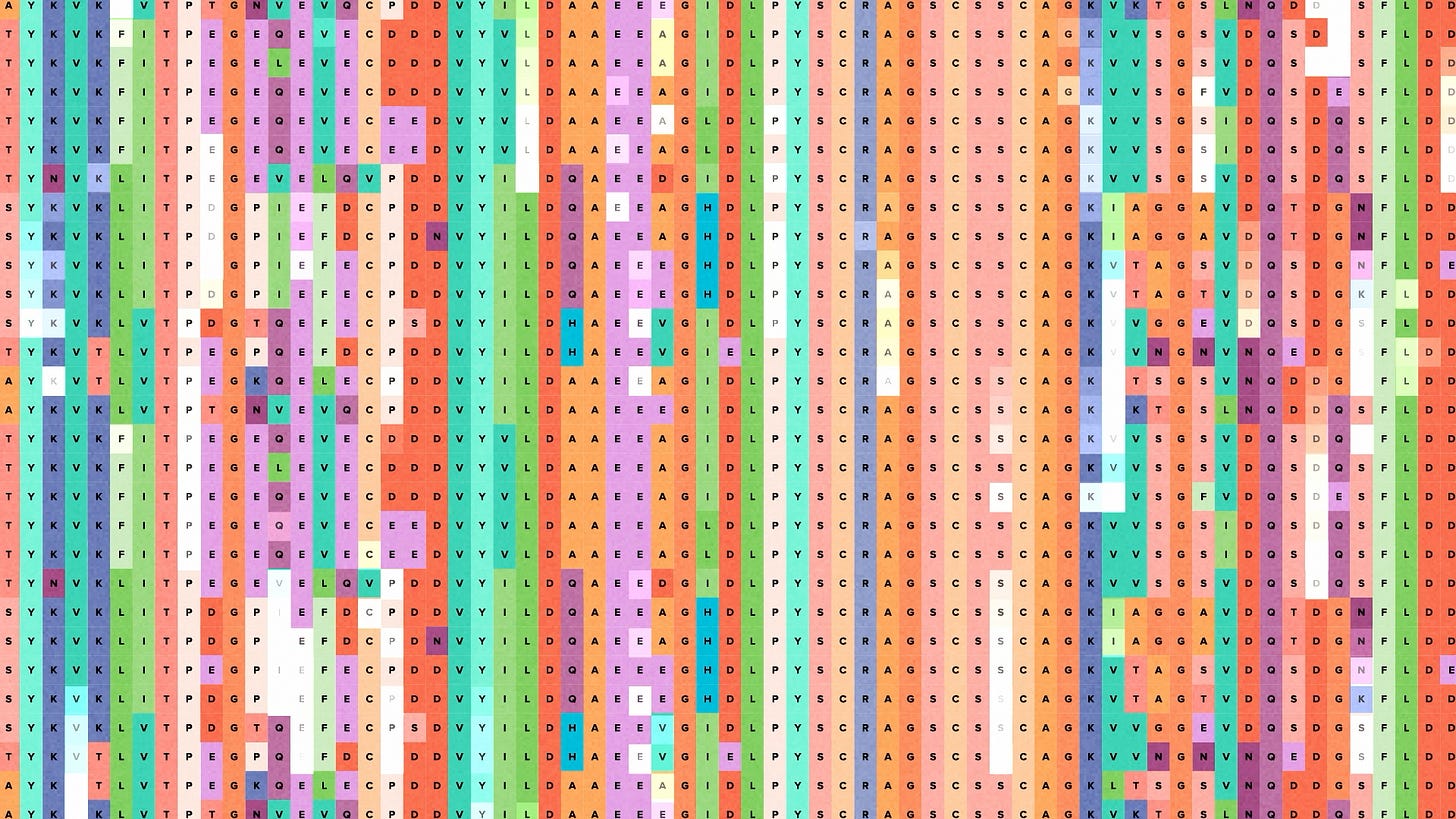
Protein misfolding is fairly common when you move a gene from one host to another. Usually proteins spontaneously fold themselves into tight little functional knots. But when you change the protein environment, that process can be less efficient. You end up with these spaghetti-like blobs that stick to everything and cause all kinds of trouble.
With the help of AI, we can design protein variants that fold better in their new host. Natural proteins tend to be good at folding, because if they weren't, evolution would eliminate them. So AI models trained on natural protein sequences can generate sequence variants that are evolution-like. With the power of the foundry, we can build and test all of those variants in real cells to see which ones express and fold efficiently.
Toxic metabolic intermediates. Generally speaking, there's a lot of chemistry going on inside a cell. In any given metabolic pathway, some molecules might include reactive groups that can cause damage.
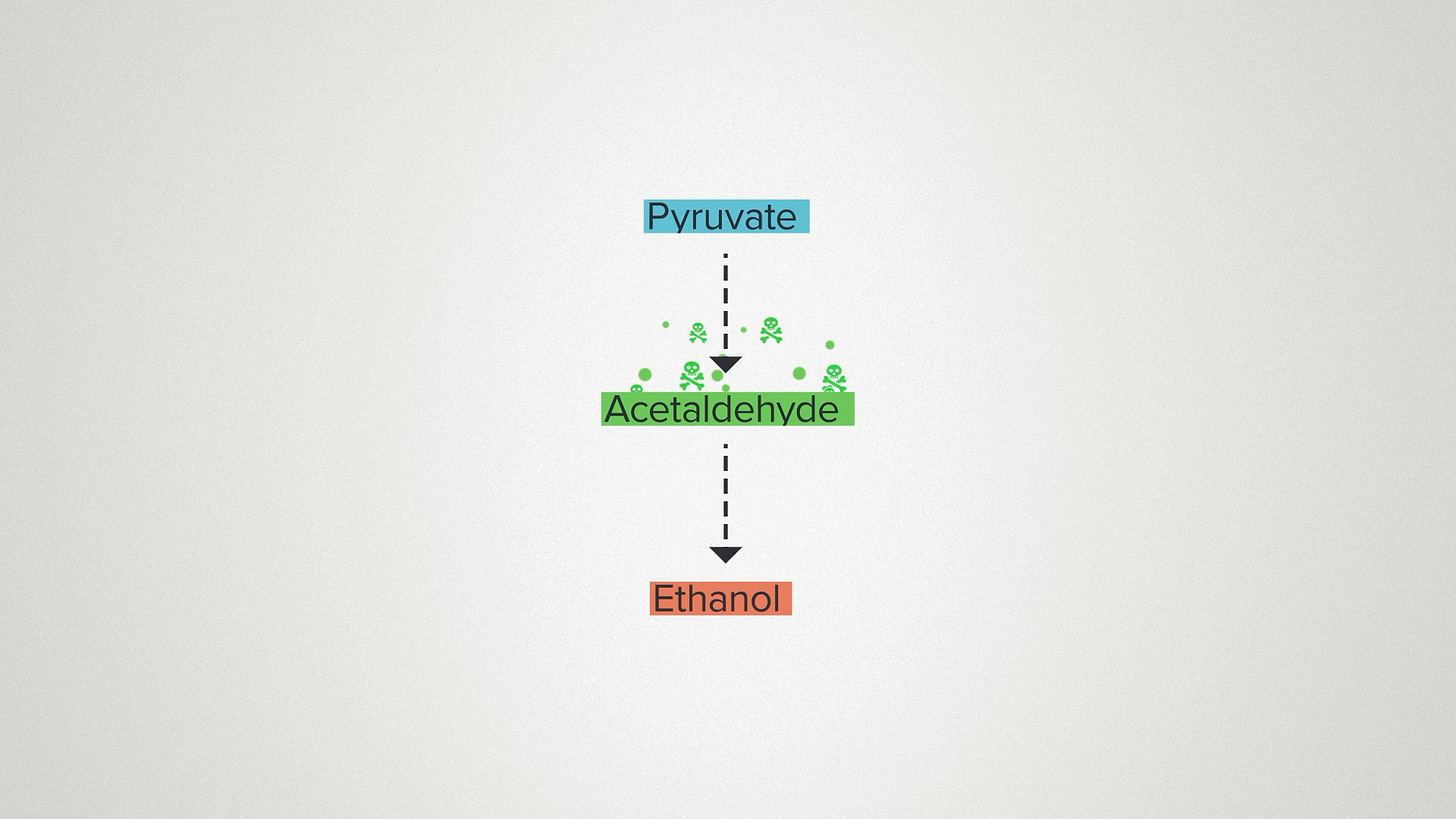
A classic example of this happens during alcoholic fermentation. The yeast metabolize glucose to pyruvate, then to acetaldehyde, and finally to ethanol. The pyruvate is fine. And the ethanol is ok, at least if you're a yeast. But the acetaldehyde in between is pretty toxic.
When this happens in a different metabolic pathway, we have a few different strategies depending on the circumstances. One solution is to move molecules through those pathways as quickly as possible. We want less of the enzyme that produces the toxic molecule and more of the enzyme that consumes it. This keeps things flowing through the pathway so the intermediate doesn't accumulate.
Another approach here might be called just doing clever chemistry. Metabolism is versatile and there are usually multiple different routes to create a compound. If a particular molecule is toxic, maybe we can mask that toxicity with a side group. Maybe we can produce a similar molecule that is easy to convert back to the desired target at a later stage. This conversion might happen later in the metabolic pathway, or during downstream processing, whichever is more efficient.
Finally, toxic products. Sometimes the thing you want to make is itself hard for cells to tolerate. Ethanol is another good example here. Yeast produce it naturally through fermentation. It also kills microbes at high concentrations. Toxic products are usually a case of "the dose makes the poison." Lots of molecules that are harmless at low concentrations can become challenging when you engineer cells to produce them at very high titer.
To solve this problem, we can take a page from the yeast playbook. Because yeast are often soaking in ethanol, they've evolved to handle it. We can similarly evolve microbial strains to tolerate higher concentrations of your product. Our tech stack in the foundry includes a platform for Adaptive Laboratory Evolution. It starts with a low concentration of the toxic product, maybe it's a solvent or an acid, and gradually brings it up over time. After 2 or 3 months of continuous automated culture, evolution will find the necessary mutations to make the host more resistant.
Toxicity makes biology hard tech because it isn't really just one problem. It includes problems that you might know about in advance, and problems that come up during the course of a project. Solving for toxicity means being ready with a whole foundry full of tools: AI, human design, evolution, and having the experimental capacity to try them all.
It isn't easy. But you know what? The cells are worth it. They're resilient, hardworking little machines. They do a lot for us, and they deserve a lot of TLC to keep them healthy and happy.